Excellence in communication science
Leading journals publish CNIC science
JACC
A drug costing less than €2 a day helps in the treatment of severely ill COVID-19 patients
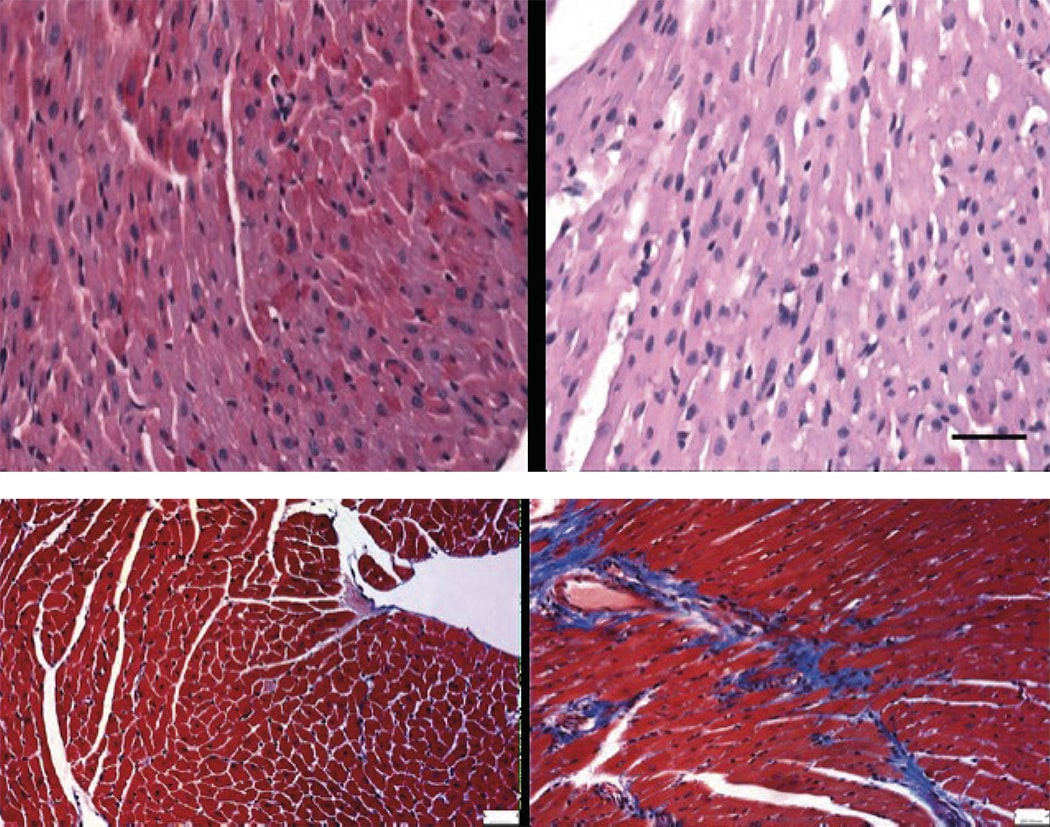
The most severe form of COVID-19 is severe respiratory failure, which requires intubation and is associated with a high mortality rate. Pulmonary infection with the SARS-CoV2 virus can progress to acute respiratory distress syndrome (ARDS), in which inflammation and neutrophil hyperactivation play a central role. There is currently a lack of therapies for ARDS associated with COVID-19.
The study was led by Dr. Borja Ibáñez, group leader of the Translational Laboratory for Cardiovascular Imaging and Therapy at the CNIC, cardiologist at the Hospital Universitario Fundación Jiménez Díaz (FJD) in Madrid, and member of the CIBERCV cardiovascular research network. The research team recently discovered that metoprolol, a well-established beta-blocker, has a highly selective effect on hyperactivated neutrophils during situations of acute stress such as a myocardial infarction. Given the central role played by neutrophils in ARDS, the team postulated that metoprolol might be an effective treatment for patients with severe COVID-19.
Madrid-COVID is a randomized clinical trial conducted in close collaboration between the CNIC and cardiology, ICU, pulmonology, and biobank services at FJD Hospital. This pilot trial examined the effect of intravenous metoprolol administration on lung inflammation and respiratory function in severe COVID-19 patients intubated after developing ARDS.
The team “randomized 20 intubated COVID-19 patients to receive intravenous metoprolol (15 mg per day over 3 days) or to a control group that did not receive metoprolol. We analyzed the inflammatory infiltrate in bronchoalveolar fluid before and after treatment and also monitored clinical progression parameters such as oxygenation and days on mechanical ventilation.” The intravenous metoprolol treatment significantly reduced neutrophil infiltration of the lungs and improved oxygenation.
Dr. Ibáñez added that “while we need to be cautious with these results of a pilot trial, we have observed that metoprolol treatment in this clinical setting is safe, is associated with a very significant reduction in lung infiltration, and appears to lead to very rapid improvements in patient oxygenation.”
The researchers therefore propose intravenous metoprolol as a “promising intervention that could improve the prognosis of severely ill COVID-19 patients.” They also emphasize that metoprolol is a safe and cheap drug (daily treatment cost below €2) that is readily available.
Joint first author Agustín Clemente-Moragón added that “the effect of metoprolol on the hyperactivation of inflammatory cells implicated in ARDS is exclusive to this beta-blocker.” In a previous experimental study, the same group recently demonstrated that other apparently similar beta-blockers have no effect on exacerbated lung inflammation.
The research team led by Dr. Ibáñez was awarded funding from the Instituto de Salud Carlos III (ISCIII) for a clinical trial to definitively demonstrate the clinical benefits of metoprolol in 350 ADRS patients admitted to 14 ICUs across Spain. The MAIDEN clinical trial will be coordinated by the cardiovascular CIBER research network and will include the participation of cardiovascular and respiratory specialists.
The study was partly funded by the European Comission (EERC-CoG grant Nº 819775) and the Ministerio de Ciencia e Innovación (MCN; ‘RETOS 2019’ grant Nº PID2019-107332RB-I00). The study was also supported by the Programa de Atracción de Talento de la Comunidad de Madrid.

Circulation Research
CNIC scientists describe one of the mechanisms
underlying cardiac hypertrophy
Scientists at the Centro Nacional de Investigaciones Cardiovasculares (CNIC) have identified a new mechanism involved in the regulation of cardiac hypertrophy. The mechanisms underlying this disease remain largely unknown, and this situation is impeding the development of effective treatments.
The findings, published in Circulation Research, may spur the development of new tools for the treatment of cardiac hypertrophy, especially in Cushing syndrome patients.
The results, explained principal investigator Dr. Enrique Lara Pezzi, who leads the Molecular Regulation of Heart Failure group at the CNIC, reveal that the “protein SRSF4 binds to and stabilizes the non-coding RNA GAS5, enabling it to block the action of the glucocorticoid receptor and thus prevent cardiac hypertrophy.”
The contractile function of the heart depends on the action of cardiomyocytes. Although in the adult heart these cells have almost no ability to divide, Dr. Lara Pezzi explained that they have a remarkable ability to adapt to the changing demands placed on the heart.
One example of this capacity is the response to aortic stenosis. “During stenosis, the aortic valve cannot open fully, and the narrowing of the opening obliges the left ventricle to pump blood with greater force. This requires the heart muscle to contract more strongly,” continued Dr. Lara Pezzi.
Although cardiomyocytes cannot divide, Dr. Lara Pezzi explained that “to increase contractile capacity, these cells become enlarged, a process known as hypertrophy.”

However, while this response is effective at first, the thickening of the left ventricular wall (cardiac hypertrophy) triggers structural changes in the heart that cause a progressive loss of contractile capacity.
Continuing lack of knowledge about the mechanisms underlying cardiac hypertrophy is impeding the development of effective treatments.
Dr. Lara Pezzi’s group, in partnership with researchers at Hospital Puerta de Hierro in Majadahonda, the Spanish Cardiovascular Research Network (CIBERCV), and the University of Frankfurt, has analyzed possible mechanisms underlying the development of this disease.
The researchers found that mice lacking the RNA-binding protein SRSF4 develop cardiac hypertrophy and have an impaired ability to relax the heart muscle, a condition known as diastolic dysfunction.
Further analysis showed that the absence of SRSF4 severely reduces the expression of the non-coding RNA GAS5. “SRSF4 binds directly to GAS5, preventing GAS5 degradation in the cell. GAS5 is an inhibitor of the glucocorticoid receptor, whose activation contributes to the development of cardiac hypertrophy,” explained Dr. Lara Pezzi.
First author Dr. Javier Larrasa clarified that “the absence of SRSF4 triggers the degradation of GAS5, and this results in activation of the glucocorticoid receptor. In contrast, overexpression of GAS5 with a viral vector inhibits the glucocorticoid receptor and reduces cardiac hypertrophy.”
The research team concludes that the identification of the SRSF4-GAS5-glucocorticoid-receptor cell signaling pathway is a major advance in the characterization of the molecular mechanisms underlying cardiac hypertrophy and could serve as the basis for new treatments.
Furthermore, the findings could be “particularly applicable to patients with Cushing syndrome, who develop cardiac hypertrophy in part through glucocorticoid receptor activation.”
An analysis of samples from Cushing syndrome patients revealed a degree of dysregulation of SRSF4 and GAS5 expression; however, the possible role of this pathway in this disease will require further research.
The study received funding from the European Union (CardioNeT-ITN-289600 and CardioNext-ITN-608027), the Ministerio de Economía y Competitividad, and the Community of Madrid (2010-BMD-2321 “Fibroteam”). Funding was also provided by the Plan Estatal de I+D+I 2013-2016 and the European Regional Development Fund “A way to build Europe” program.
Scientific Reports
3D mapping of post-infarction scarring increases the prognostic potential of cardiac magnetic resonance imaging
A multidisciplinary team of scientists based at the Universidad de Valladolid and the Centro Nacional de Investigaciones Cardiovasculares (CNIC), has developed a highly efficient method for identifying the 3-dimensional features of the scar tissue formed after a myocardial infarction. The study was carried out in partnership with scientists and clinicians at Hospital Clínico San Carlos, Hospital Universitario La Paz, Fundación Jiménez Díaz, Hospital Ruber Juan Bravo Quironsalud, Universidad Politécnica de Madrid, Centro de Supercomputación de Barcelona, Philips Healthcare Iberia, CIBERCV and CIBERBBN.
The new method allows 3-dimensional transmural (across the ventricular wall) mapping of scar tissue in the infarcted muscle. Transmural mapping of the infarcted tissue enables highly detailed characterization of the morphology of the damaged tissue and provides an accurate measure of infarct size relative to myocardial wall thickness, a parameter known as transmurality.
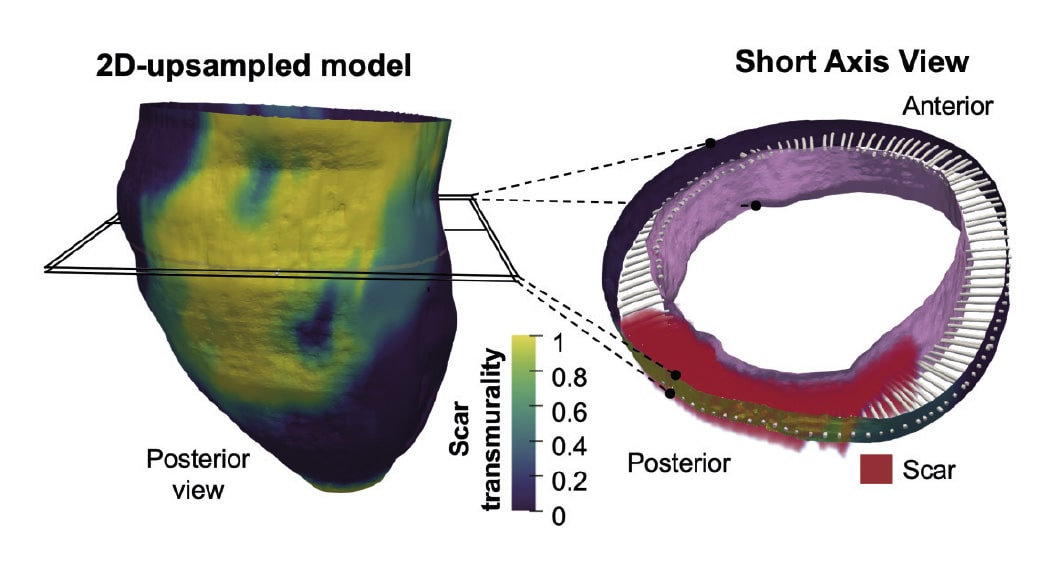
According to CNIC scientist David Filgueiras, “a major advantage of this new method is its full compatibility with standard cardiac magnetic resonance (CMR) sequences that take just a few minutes to acquire. This approach can thus significantly shorten the time needed for image acquisition, easing access to in-demand nuclear magnetic resonance scanners.”
“This novel methodology may provide an efficient approach in clinical practice after manual or automatic segmentation of myocardial borders in a small number of conventional 2D slices and automatic scar detection,” write the authors in an article published in Scientific Reports.
The results of the study show that low scar transmurality on CMR (below 10% of ventricular wall thickness for 3D sequences or 20% for 2D sequences) is associated with the clinical presentation of tachycardias involving infarcted ventricular tissue, known as ventricular tachycardias.
Describing the results study first authors Susana Merino, of Valladolid University, and Lilian Karina Gutiérrez, of the CNIC, said that the results reveal a significant correlation between low scar transmurality and the cardiac frequency of spontaneous ventricular tachycardia episodes.
The results also show that patients with low scar transmurality values had a higher probability of ventricular tachycardia recurrences during long-term follow-up.
The new method is especially promising because it uses conventional 2D delayed gadolinium-enhanced CMR sequences and requires only a limited number of slices. This technology is available at all centers that carry out CMR studies, and the method does not require 3D CMR studies.
The method was developed through technical, experimental, and clinical collaboration under the umbrella of a specific partnership between Valladolid University and the CNIC. Joint lead author Carlos Alberola explained that the technical advances of the method are built on procedures developed in the Image Processing Laboratory at Valladolid University, located in the Escuela Técnica Superior de Ingenieros de Telecomunicación.
“The first of these advances is a method for interpolating images that preserves topology. This allows high-resolution 3D images to be generated with a high level of precision from the images obtained with conventional CMR procedures.”
“A second advance is a mathematical method for characterizing fibrous tissue formed in the myocardial wall after an infarction. This tool provides a measure of the full 3D morphology of the scar relative to the thickness of the myocardium, a parameter known as transmurality.” The method uses a procedure based on partial differential equations to provide point-wise correspondences between the endocardium and the epicardium. These correspondences allow the definition of multiple indicators of the extent of the infarction.
The experimental arm of the study involved input from the Advanced Development in Arrhythmia Mechanisms and Therapy group at the CNIC, led by Dr. David Filgueiras. This group provided expertise in experimental models of myocardial infarction, essential for validating the methodology.
The CNIC laboratory also coordinated the study of the clinical application of the new method, in partnership with a multidisciplinary team of experts in the diagnosis and treatment of complex cardiac arrhythmias.
The study received funding from the Ministerio de Ciencia e Innovación (TEC2017-82408-R y PID2019-109329RB-I00); Fondo Europeo de Desarrollo Regional (CB16/11/00458); Heart Rhythm Association de la Sociedad Española de Cardiología; Fundación Interhospitalaria para la Investigación Cardiovascular (FIC); Fundación Eugenio Rodríguez Pascual; programa H2020 de la Unión Europea; Ministerio de Asuntos Económicos y Transformación Digital, and Fundación Carolina-BBVA.
Science
A rapid mechanism for muscle self-repair independent of stem cells
Muscle is known to regenerate through a complex process that involves several steps and relies on stem cells. Now, a new study led by researchers at Universitat Pompeu Fabra (UPF), Centro Nacional de Investigaciones Cardiovasculares (CNIC) / CIBERNED and Instituto de Medicina Molecular João Lobo Antunes (iMM, Portugal), published on 15 October in the journal Science, describes a new mechanism for muscle repair after physiological damage relying on the rearrangement of muscle fibre nuclei, and independently of muscle stem cells. This protective mechanism paves the way to a broader understanding of muscle repair in physiology and disease.
Skeletal muscle tissue, the organ responsible for locomotion, is formed by cells (fibres) that have more than one nucleus, an almost unique feature in our body. Despite the plasticity of these fibres, their contraction can be associated with muscle damage. William Roman, first author of the study and researcher at UPF, explains: “Even in physiological conditions, regeneration is vital for muscle to endure the mechanical stress of contraction, which often leads to cellular damage.” Although muscle regeneration has been investigated in depth in recent decades, most studies centred on mechanisms involving several cells, including muscle stem cells, which are required when extensive muscle damage occurs.”
“In this study we found an alternative mechanism of muscle tissue repair that is muscle-fibre autonomous,” says Pura Muñoz-Cánoves, ICREA professor and principal investigator at UPF and the CNIC, and study leader. Even in physiological conditions, regeneration is vital for muscle to endure the mechanical stress of contraction, which often leads to cellular damage.
Researchers (including Antonio Serrano (UPF) and Mari Carmen Gómez-Cabrera (Universitat de València e INCLIVA) used different in vitro models of injury and models of exercise in mice and humans to observe that upon injury, nuclei are attracted to the damage site, accelerating the repair of the contractile units. Next, the team dissected the molecular mechanism of this observation: “Our experiments with muscle cells in the laboratory showed that the movement of nuclei to injury sites resulted in the local delivery of mRNA molecules. These mRNA molecules are translated into proteins at the site of injury to act as building blocks for muscle repair,” explains William Roman. “This muscle fibre self-repair process occurs rapidly both in mice and in humans after exercise-induced muscle injury, and thus represents a time- and energy-efficient protective mechanism for the repair of minor lesions,” adds Pura Muñoz-Cánoves.
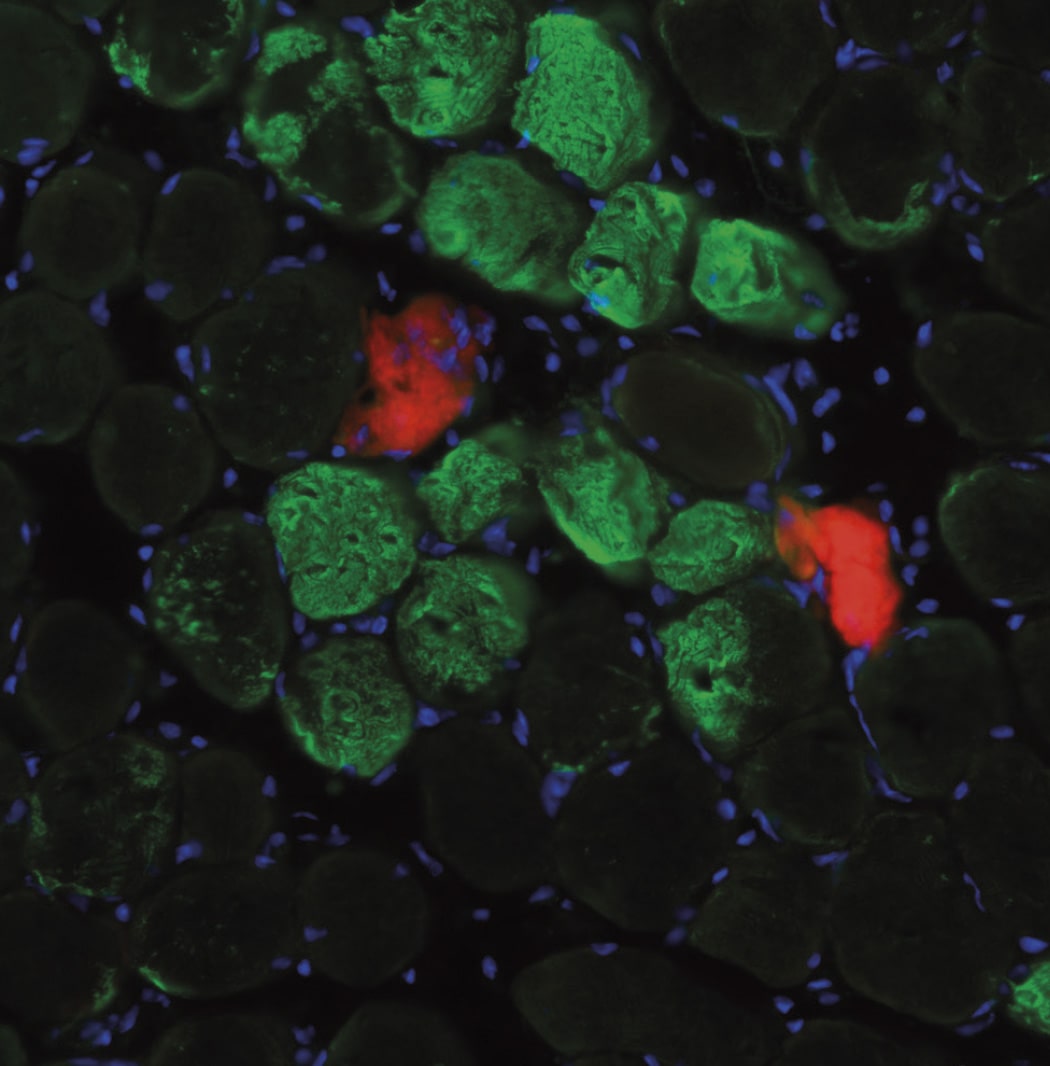
In addition to its implications for muscle research, this study also introduces more general concepts for cell biology, such as the movement of nuclei to injury sites. “One of the most fascinating things about these cells is the movement during the development of their nuclei, the biggest organelles inside the cell, but the reasons why nuclei move are largely unknown. Now, we have shown a functional relevance for this phenomenon in adulthood during cell repair and regeneration,” says Edgar R. Gomes, group leader at the Instituto de Medicina Molecular and a professor at the Faculty of Medicine at the University of Lisbon, who co-led the study.
This finding constitutes an important advance in the understanding of muscle biology, in physiology (including exercise physiology) and muscle dysfunction.
On the importance of these discoveries, Pura Muñoz-Cánoves, Antonio Serrano and Mari Carmen Gómez-Cabrera agree that: “This finding constitutes an important advance in the understanding of muscle biology, in physiology (including exercise physiology) and muscle dysfunction.”
The work was conducted at UPF/CNIC/CIBERNED and at the iMM in collaboration with the University of Valencia/INCLIVA. This study was funded by the European Research Council, Association Française contre les Myopathies, the European Molecular Biology Organization, the Human Frontiers Science Programme, and the Spanish Ministry of Science.
Circulation
It’s never too late to treat progeria
Scientists at the Centro Nacional de Investigaciones Cardiovasculares Carlos III (CNIC) and the Spanish Cardiovascular Research Network (CIBERCV), led by Dr. Vicente Andrés, have generated the HGPSrev experimental mouse model. This is the first animal model to develop Hutchinson-Gilford Progeria Syndrome (HGPS) and also allow its suppression through the controlled regulation of the expression of progerin, the aberrant protein that causes the disease. Using the new model, the researchers have demonstrated that it is never too late to treat HGPS.
The study, published in Circulation, also establishes that the cardiovascular alterations and early death associated with HGPS can be prevented with treatments specifically targeting cells of the cardiovascular system.
HGPS is an ultra-rare genetic disease that affects fewer than 400 children worldwide and for which there is no known cure. The disease is caused by a mutation in the LMNA gene and is characterized by accelerated aging and death in the second decade of life, principally due to cardiovascular complications derived from atherosclerosis.

In the absence of mutations, LMNA encodes type A lamin proteins (Lamins A and C). The mutation found in HGPS patients results in the synthesis of progerin, a mutant protein that provokes multiple molecular and cellular alterations in the tissues where it accumulates, causing their life to pass at an highly accelerated rate, where minutes are hours and hours are lost days.
Now, thanks to the HGPSrev mouse generated by the CNIC Molecular and Genetic Cardiovascular Pathophysiology group, the research team has managed to suppress progerin expression and reestablish lamin A expression in mice of different ages, both throughout all body tissues and in specific cell types. The characterization of the animal model was carried out with the participation of researchers at Queen Mary University of London.
Joint first authors Drs. Amanda Sánchez López and Carla Espinós Estévez explained that while some palliative therapies are effective in animal models and are the subject of clinical trials, their therapeutic benefit is very limited. “A true cure would require the elimination of the culprit mutation,” commented Dr. Sánchez López. However, this is not yet possible, and progeria is only diagnosed once the first symptoms have already appeared. “We therefore sought to reverse symptoms once they are already present and to determine how long treatment could be delayed and still have a beneficial impact,” explained Dr. Espinós Estévez.
The extent to which the damage caused by progerin can be reversed is currently not known, and patients often do not start to receive treatment until symptoms are quite advanced. The investigators therefore addressed a key question: Can the progression of HGPS be stopped or slowed if treatment commences when the disease is advanced, or does therapeutic benefit depend on starting treatment early, when symptoms are mild?
Another important question is how treatment should be targeted. Progerin is expressed in many tissues, but it was not known if treatment needs to be directed at all affected cells or if it would be effective if targeted at a specific cell type. To answer these questions, Dr. Andrés’s team used CRISPR-Cas9 technology to generate HGPSrev mice.
The results published in Circulation show that HGPSrev mice develop the major features of the human disease, including growth retardation, lipodystrophy, cardiovascular alterations, and early death.
The investigators also showed that elimination of progerin and restoration of lamin A expression increased life expectancy by 84.5% in HGPSrev mice with very mild symptoms and, moreover, extended lifespan by 6.7% even in mice with very advanced symptoms.
These results establish not only that starting treatment when symptoms are mild has a huge positive impact, but also that treatment can be beneficial no matter how late it is started.
“We managed to prevent vascular alterations and normalize survival in progeric mice by eliminating progerin expression and restoring lamin A expression specifically in vascular smooth cells and cardiomyocytes, even though other cell types remained diseased,” explained Dr. Andrés.
The investigators conclude that these results “could contribute to the design of future clinical treatments, given that they suggest that strategies exclusively targeting the cardiovascular system could have a very significantly beneficial effect on patients’ life quality and expectancy.”
The study received funding from the Ministerio de Ciencia e Innovación (MCIN)/Agencia Estatal de Investigación (AEI)/10.13039/501100011033 (grants SAF2016-79490-R, PID2019-108489RB-I00, SVP-2014-068334, FJCI-2017-33299); Instituto de Salud Carlos III (ISCIII; grant AC17/00067-TREAT-HGPS), an E-Rare Joint Transnational call project, European Union Horizon 2020 Framework Programme 2017); the Fondo Europeo de Desarrollo Regional (“a way to build Europe”); the Wellcome Trust (grant 098291/Z/12/Z); the Comunidad Autónoma de Madrid (grant 2017-T1/BMD-5247); the Asociación Apadrina la Ciencia-Ford España-Ford Motor Company Fund; and the Fundación “la Caixa”.
Science Advances
CNIC scientists identify the essential role of a gene in placental development
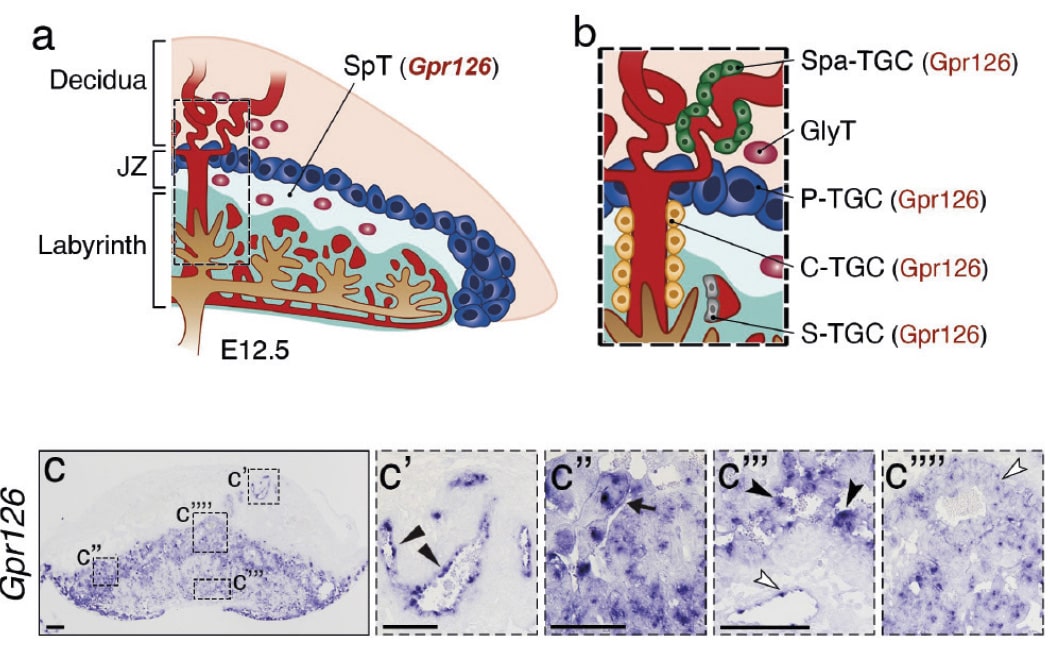
Scientists working with an experimental mouse model at the Centro Nacional de Investigaciones Cardiovasculares (CNIC) have identified the essential role of the gene GPR126 in the development of the placenta during pregnancy.
The results, published in Science Advances, show that GPR126 (adhesion G protein-coupled receptor 126) is essential for the development of a specific placental cell type that regulates the remodeling of the uterine vasculature. Cardiac defects in mouse Gpr126 mutants are secondary to placental defects, reflecting the intimate relationship between the placenta and the fetal heart.

There is evidence that GPR126 may play a similar role in placental development in humans. The babies of women who carry mutations in GPR126 die during gestation or soon after birth, and 30% of these women develop preeclampsia. This pregnancy complication, which affects 5-8% of pregnancies in the general population, is characterized by high blood pressure that damages both mother and fetus and can result in fetal death.
Experiments in animal models have shown that GPR126 is required for the maturation of the peripheral nervous system (PNS), the formation of bone and cartilage, and the development of the inner ear. In humans, mutations in GPR126 are associated with skeletal malformations and muscle cramping in the limbs.
The CNIC Intercellular Signaling in Cardiovascular Development and Disease group, led by Dr. José Luis de la Pompa, initially identified GPR126 as a gene regulated by the NOTCH signaling pathway (a highly conserved intercellular signaling system in animals) during heart development.
This suggested that GPR126 might influence the proliferation and differentiation of cardiomyocytes (heart muscle cells) in the developing heart.
Other groups had already proposed a requirement for GPR126 in heart development in mice and zebrafish, but research had not provided conclusive evidence to confirm this hypothesis.
In the new study published in Science Advances, the CNIC team demonstrate using genetic techniques that GPR126 is not necessary for heart development in the mouse but plays an essential role in the formation of the placenta, a process known as placentation.
Explaining the results, Dr. De la Pompa said that “We observed that whole-body inactivation of GPR126 in mice caused thinning of the walls of the heart and resulted in embryonic death. However, when we inactivated the gene specifically in the heart, embryonic development was unaffected and heart function unaltered.”
The investigators also found that the lethal effect of whole-body GPR126 deletion “was not reversed by specific re-expression of GPR126 in the heart, indicating that embryonic death in the mutants was not due to a defect in cardiac development,” explained first author Rebeca Torregrosa. Further studies in the zebrafish model confirmed that GPR126 “is not involved in heart development.”
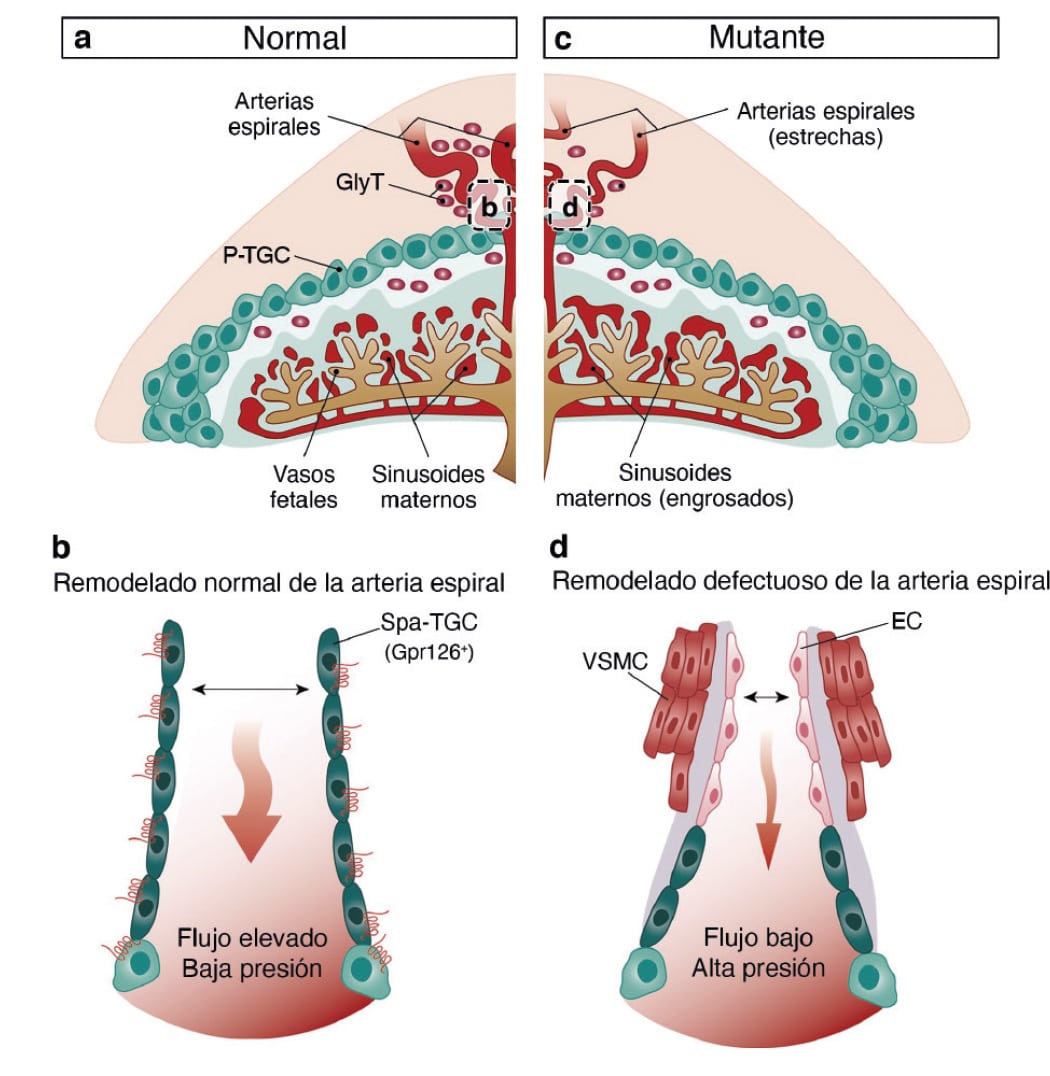
During embryonic development, GPR126 is expressed in giant trophoblast cells, a cell type specific to the placenta. “These cells,” indicated De la Pompa, “are vitally important for the implantation of the embryo and the maintenance of pregnancy.”
The research team demonstrated that GPR126 inactivation in the embryo is compatible with survival if the placenta retains at least one normal copy of the GPR126 gene. However, inactivation of the gene in both the embryo and the placenta causes embryonic death.
“One of the crucial steps in placental development is the remodeling of the maternal arteries. Known as the spiral arteries, these vessels increase their diameter to increase blood flow to the embryo. Defects in this process are associated with pregnancy complications such as preeclampsia, restricted fetal growth, and even miscarriage,” said José Luis de la Pompa.
The study shows that trophoblast GPR126 is necessary for the expression of specific proteases involved in spiral artery remodeling, which is essential for the viability of the fetus.
Based on these results, the investigators herald mice lacking GPR126 as an experimental model for studying spiral artery remodeling and preeclampsia. This animal model, moreover, has possible clinical applications in preimplantation genetic diagnosis.
The study was funded by the cardiovascular research network CIBER CV (PID2019-104776RB-I00, CB16 / 11/00399), TERCL (RD16 / 0011/0021) of the Ministerio de Ciencia, Innovación y Universidades (MCIU), BBVA Foundation, “la Caixa” Foundation, EMBO fellowship, Boehringer Ingelheim Fonds, and the Programa de Atracción de Talento of the Comunidad de Madrid (Ref.2016T1 / BMD1540).
Frontiers IN Immunology
CNIC scientists show that trained innate immunity protects against SARS-CoV-2 and increases the immunogenicity of COVID-19 vaccines
COVID-19 vaccines provide the best weapon against the pandemic caused by the SARS-CoV-2 coronavirus. But these vaccines have also highlighted the need for effective and fast acting responses against future viral threats. This can be achieved by activating–or training–a part of the body’s natural defense system called innate immunity. This is the conclusion of a multicenter study coordinated by Spanish scientists Carlos del Fresno, of the Instituto de Investigación Sanitaria del Hospital Universitario La Paz (IdiPAZ), Juan García Arriaza and Mariano Esteban of the Centro Nacional de Biotecnología (CNB)/Consejo Superior de Investigaciones Científicas (CSIC), and David Sancho of the Centro Nacional de Investigaciones Cardiovasculares (CNIC).
In the study, published in Frontiers in Immunology, the team demonstrate that immune therapy with the MV130 dead bacterial preparation, produced by the Spanish biotech company Inmunotek S.L. in Alcalá de Henares, protects against SARS-CoV-2 infection in susceptible mice. The study was carried out at the Centro de Investigación en Sanidad Animal (CISA), part of the Instituto Nacional de Investigación y Tecnología Agraria y Alimentaria (INIA-CSIC), in Madrid. The mortality rate after SARS-CoV-2 infection was significantly lower in mice that had previously received the MV130 immune therapy. This treatment trains the innate immune system by introducing epigenetic changes into innate immune cells, as demonstrated in previous studies in Dr. Sancho’s laboratory.
The research team investigated whether this immune therapy might improve the immune response generated in response to subsequent vaccination against COVID-19. In experiments carried out at the CNIC, mice treated with MV130 or a control preparation were innoculated with one of two types of vaccine: one based on the CSIC MVA-CoV2-S vaccine (generated by Drs. García Arriaza and Esteban) and another based on recombinant protein S with adjuvant. Two vaccine administration routes were tested: intramuscular injection and the intranasal route.
“Animals treated with MV130 before vaccination, which therefore had a trained innate immune system, had a better immune responses to vaccination,” explained Carlos del Fresno. “MV130 treatment increased the activation of cytotoxic CD8 T cells, which eliminate infected cells, as well as the levels of IgA-class antibodies to SARS-CoV-2 protein S in mucous membranes.”
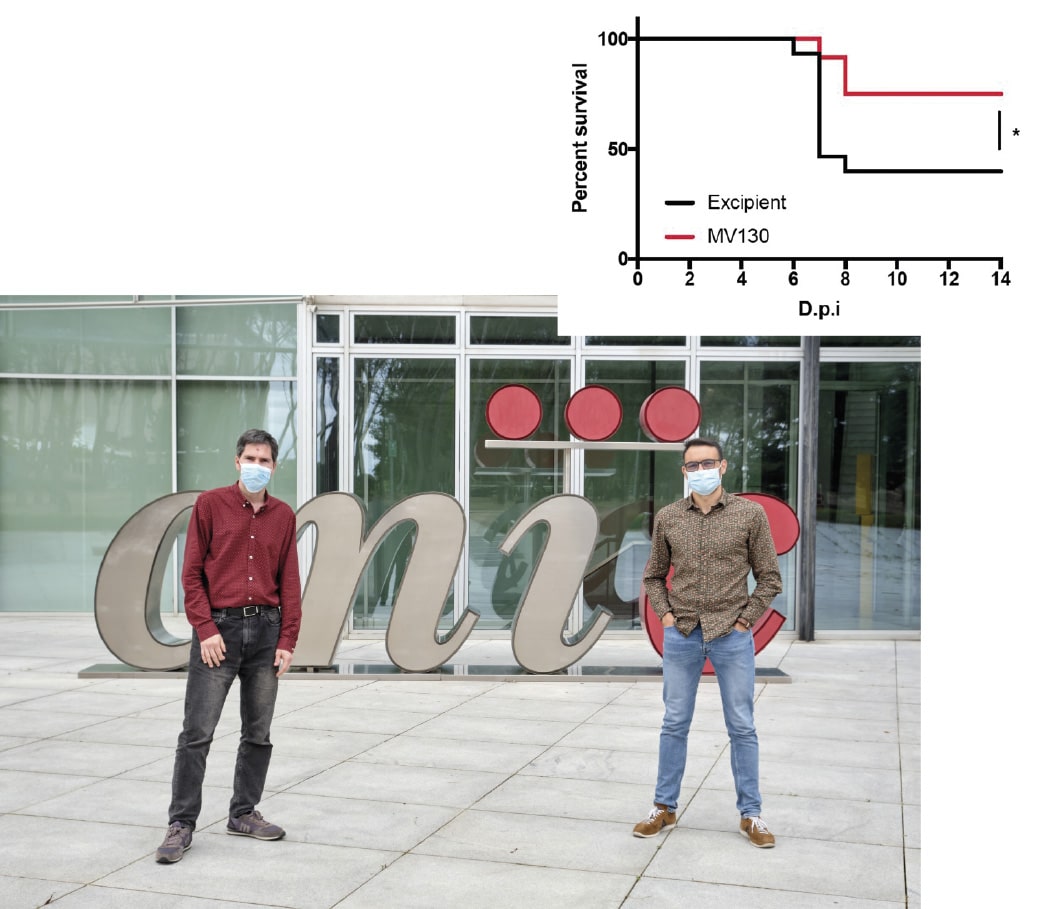
Summarizing the impact of the findings, Juan García Arriaza said: “The study shows that immune therapy with MV130 directly protects against death from SARS-CoV-2 infection and shows the ability of trained innate immunity to improve immune responses generated by COVID-19 vaccines.”
The authors conclude that MV130 immune therapy will improve vaccine efficacy and immunogenicity, especially in vulnerable population groups or against variants against which vaccines are less effective, thus contributing to better community protection against COVID-19.
The study was supported by funding from the Fondo Solidario Juntos Banco Santander for Dr. David Sancho’s research at the CNIC and the Fondo COVID-19 Instituto de Salud Carlos III for Dr. Juan García Arriaza’s work. Other supporting bodies were the Fundación Asociación Española Contra el Cáncer (AECC); Fondo Supera COVID-19 (Crue Universidades-Banco Santander); Fundación ‘la Caixa’; Programa Horizonte 2020 and Beca Marie Skłodowska-Curie de la Unión Europea; an EMBO fellowship; Comunidad de Madrid and Fondo Social Europeo y Desarrollo Regional Europeo; Consejo de Investigación Europea (ERC-2016-Consolidator Grant 725091); Agencia Estatal de Investigación (PID2019-108157RB); Comunidad de Madrid; Inmunotek S.L., and Fundació La Marató de TV3.
PLoS Biology
CNIC scientists identify two proteins essential for postnatal cardiac metabolism

Scientists at the Centro Nacional de Investigaciones Cardiovasculares (CNIC) have identified essential roles for two proteins in cardiac metabolism after birth. The study, published in PLoS Biology, shows that forced premature activation of these proteins in the mouse heart during the first days of life causes irreversible damage and alters whole-body metabolism, leading to diabetes and impaired thermoregulation later in life.
Lead investigator Dr. Guadalupe Sabio explained that, fortunately, the study shows that “these effects could in principle be treated through a change in diet.”
During fetal development and the first days after birth, the heart derives most of its energy from the metabolism of glucose, stored in the form of glycogen. But first author Ayelén Santamans explained that, soon after birth, the rapid growth of the heart increases the organ’s energy demands, requiring the heart to become “much more efficient at obtaining energy.”
The PLoS Biology study shows that the proteins p38γ and p38δ are activated in the heart shortly after birth and reduce the activity of the enzyme responsible for glycogen synthesis. This precipitates a metabolic change in the heart, which begins to use fatty acids as its main energy source.
Perturbation of cardiac metabolism by forced premature p38γ and p38δ expression during the postnatal period causes irreversible damage that manifests in adulthood as insulin resistance, glucose intolerance, and an impaired ability to maintain body temperature.
But the new study shows that, since the problem is an insufficient supply of energy to the heart, the damage can be repaired by a change in diet. To demonstrate this, the researchers fed female mice a high-fat diet during pregnancy and lactation.
Newborn mice in these experiments had no cardiac damage and did not go on to develop diabetes symptoms even when p38γ and p38δ expression was prematurely activated.
The study is the first to show that heart metabolism in the postnatal period determines whole-body metabolism. “Our results show that the gradual increase in p38γ and p38δ activity is tightly regulated and that premature activation produces an energy deficit that is deleterious both for the heart and for the metabolism of the rest of the body,” said Dr. Sabio.
The scientists think that both p38γ and p38δ might underlie some congenital cardiometabolic diseases that currently have no known cause, suggesting that dietary supplementation could be a valid treatment for these conditions.
The study was supported by the following funding bodies: EFSD; Lilly European Diabetes Research Programme; Ministerio de Ciencia, Innovación y Universidades; Comunidad de Madrid; Fundación Jesús Serra; Instituto de Salud Carlos III; and Fundación “la Caixa”.